The scourge of counterfeit and illegal medicines in Africa
Counterfeit drugs and the distribution of illegal pharmaceuticals represent a major threat to public health in sub-Saharan Africa. This situation is exacerbated by inadequate local infrastructures, particularly as regards drug quality control laboratories.
Overview of counterfeit medicines in sub-Saharan Africa
According to the World Health Organization (WHO), around 10% of medical products in circulation in low- and middle-income countries are either substandard or falsified. In sub-Saharan Africa, this proportion can be as high as 20%, exposing populations to ineffective or even dangerous treatments. Anti-malarial drugs and antibiotics are among the most frequently counterfeited medicines, exacerbating public health problems and contributing to the development of treatment resistance.
Between January 2017 and December 2021, at least 605 tonnes of medical products were seized in West Africa during international operations, illustrating the scale of the traffic in falsified medicines in the region.
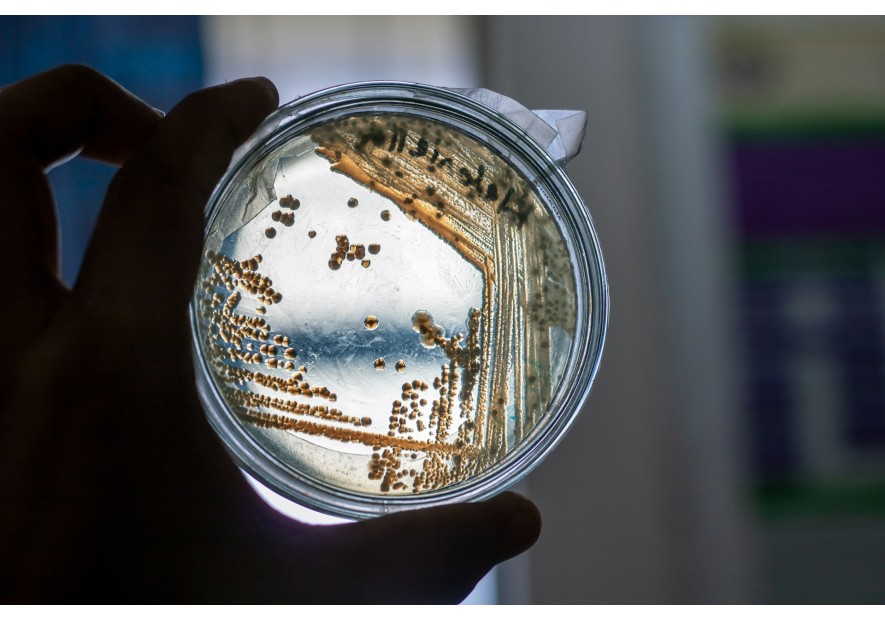
Local investigation capabilities and control laboratories
Combating counterfeit drugs requires a robust quality control infrastructure. However, in sub-Saharan Africa, national drug quality control laboratories (NDCLs) often face major challenges, including a lack of qualified human resources, adequate equipment and sufficient funding. In 2017, only six laboratories in Africa were WHO pre-qualified, and none were located in Francophone West Africa.
Initiatives have been put in place to strengthen these capacities. For example, the CORAQ-Lab project, launched in 2018, aims to improve the performance of LNCQMs in Benin, Burkina Faso, Mauritania and Niger, so that they reach the minimum standards required for a WHO prequalification application.
Inadequate local infrastructure for effective control
Despite these efforts, local infrastructures remain insufficient to ensure effective quality control of medicines on the African market. Laboratories often lack trained staff, modern equipment and quality management systems in line with international standards. This situation is compounded by often inadequate pharmaceutical regulations and limited market surveillance, allowing counterfeit medicines to proliferate.
Outlook and recommendations
To strengthen the fight against counterfeit medicines in sub-Saharan Africa, it is essential to:
- Strengthen national laboratory capacities: Invest in staff training, the acquisition of modern equipment and the implementation of quality management systems in line with international standards.
- Harmonize pharmaceutical regulations: Promote regional collaboration to establish common standards and facilitate the exchange of information between countries.
- Raise public awareness: Inform the public about the dangers of counterfeit medicines and encourage the purchase of medicines only from reliable sources.
- Strengthen international cooperation: Collaborate with international organizations, governments and private players to share resources, technologies and best practices.
In conclusion, the fight against counterfeit drugs in Sub-Saharan Africa requires a global approach involving the strengthening of local infrastructures, the implementation of effective regulations and increased international cooperation to protect the health of populations.
Check My Med - Participatory Science Check My Med - Participatory Science
Join Pharmanalyse in the fight against counterfeit and illegal drugs! Order our drug analysis and contribute to knowledge about the quality of medicines on the market. Our laboratory analyzes your drug, studies the data and publishes the results in a summary report accessible to the public....
Price €49.00


Leave a comment