Laboratory Analysis of Ketoprofen: A Necessity for Patient Safety

Among the wide range of anti-inflammatory drugs, ketoprofen plays a key role in relieving acute and chronic pain. However, the rise of e-commerce and informal supply chains has opened the door to an invisible threat: counterfeiting. In the face of this risk, laboratory analysis of ketoprofen stands as the only scientific safeguard capable of distinguishing a life-saving medication from an ineffective—or even dangerous—counterfeit.
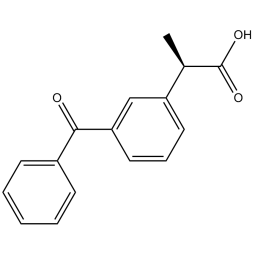
Ketoprofen: a powerful drug
Ketoprofen is a chemical compound belonging to the therapeutic class of nonsteroidal anti-inflammatory drugs (NSAIDs), and more specifically to the group of propionic acid derivatives.
Mechanism of action
Its mechanism of action is based on the inhibition of cyclooxygenase enzymes (COX-1 and COX-2). By blocking these enzymes, ketoprofen reduces the synthesis of prostaglandins, the chemical mediators responsible for inflammation, pain, and fever.
Dosage and dosage forms
Ketoprofen is known for its fast-acting effects. It is available in several forms:
- Oral route (tablets, capsules): Typically dosed at 100 mg or 150 mg, with a usual daily dose not exceeding 200 mg for adults.
- Topical route (gels): For localized joint or muscle pain.
- Injectable: Reserved for severe pain in a hospital setting or post-surgery.
The risks associated with counterfeit or substandard ketoprofen
Using a medication of questionable origin without first having it tested for ketoprofen in a laboratory exposes the patient to serious risks. Counterfeiting is not limited to simple "imitation"; it takes several concerning technical forms.
Under-dosing or the absence of the active ingredient
This is the most common scenario. An underdosed medication fails to achieve the plasma concentration necessary to inhibit COX-2. The patient suffers unnecessarily, inflammation progresses, and in the case of chronic conditions such as arthritis, irreversible tissue damage may occur. The complete absence of the active ingredient turns the medication into a mere placebo, which is dangerous due to a failure to provide proper care.
The presence of impurities and synthetic residues
The manufacture of ketoprofen requires precise chemical steps. In clandestine synthesis, the purification steps are often skipped. This can result in:
- Residues of toxic organic solvents.
- Heavy metals from catalysts.
- Inactive or toxic isomers (synthesis impurities).
Toxic excipients
To mimic the texture or color of an original tablet, counterfeiters sometimes use industrial substances not intended for human consumption, such as chalk, industrial talc, or dyes containing heavy metals, which can cause kidney or liver failure.
Pharmanalyse: The Precision of HPLC-UV at Your Service
Why choose Pharmanalyse for laboratory analysis of ketoprofen? Because trust is no substitute for analytical evidence.
The HPLC-UV method: the gold standard
Pharmanalyse uses High-Performance Liquid Chromatography coupled with Ultraviolet detection (HPLC-UV). This is the global gold standard in the pharmaceutical industry and in pharmacopoeias (USP, EP, BP).
Pharmanalyse's laboratory protocol for the analysis of ketoprofen is based on two pillars:
- Identification: We verify that the molecule in the sample has exactly the same retention time and UV spectrum as the pure ketoprofen standard.
- Quantification: By integrating the area under the chromatographic peak, we determine whether the assay result is accurate (e.g., 100 mg declared = 100 mg actual).
Expert opinion: "Only laboratory analysis can verify the identity of an active pharmaceutical ingredient and quantify its concentration in a drug. Visual or organoleptic tests are completely ineffective against modern counterfeiting techniques."
Why choose Pharmanalyse?
Whether you are a distributor looking to verify a batch, an NGO operating in sensitive areas, or an individual with concerns about an online purchase, Pharmanalyse’s laboratory analysis of ketoprofen provides you with a definitive, accurate, and legally binding answer.
In short, laboratory testing of ketoprofen ensures that your medication is a tool for managing pain, not a risk to your health. In an age when counterfeits are everywhere, analytical science is your only ally.
Ketoprofen - Profenid - Ketum - Actron Analysis Ketoprofen - Profenid - Ketum - Actron Analysis
Laboratory analysis of drugs to certify the presence of Ketoprofen and measure the quantity of Ketoprofen. The level of uncertainty of the Ketoprofen content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%). Follow the instructions to send your...
Price €149.00Check My Med - Participatory Science Check My Med - Participatory Science
Join Pharmanalyse in the fight against counterfeit and illegal drugs! Order our drug analysis and contribute to knowledge about the quality of medicines on the market. Our laboratory analyzes your drug, studies the data and publishes the results in a summary report accessible to the public....
Price €49.00


Leave a comment