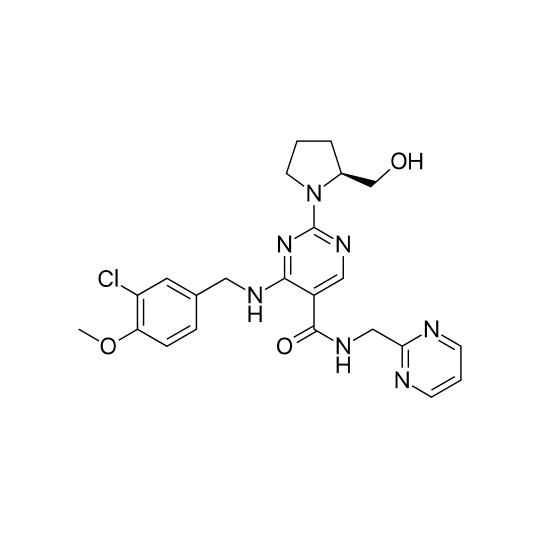
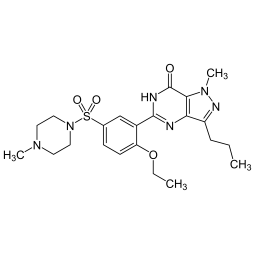
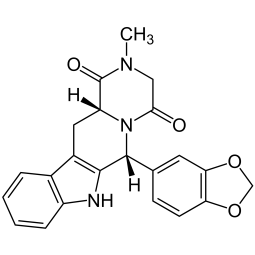
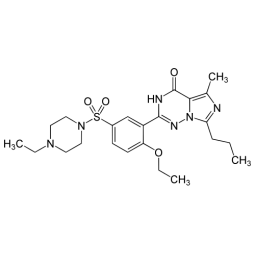
Sildenafil - Viagra - Kamagra - Reviato - Silagra Analysis Sildenafil - Viagra - Kamagra - Reviato - Silagra Analysis
Laboratory analysis of drugs to certify the presence and measure the quantity of Sildenafil (Viagra Kamagra). The level of uncertainty of the Sildenafil content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%). Follow the instructions to send...
Price €149.00