Atorvastatine - Lipitor - Tahor - Totalip Analysis
Laboratory analysis of drugs to certify the presence of atorvastatin and measure the quantity of this active ingredient.
The level of uncertainty of the atorvastatin content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%).
Follow the instructions to send your drug to our laboratory for analysis.
Results within 15 working days of receipt of sample.
Atorvastatin is a drug used primarily to reduce cholesterol and prevent cardiovascular disease.
Atorvastatin is the active ingredient in Tahor, Lipitor and many generics marketed by various pharmaceutical companies.
Consuming medicines ordered from dubious or illegal sites can be hazardous to your health. It is essential to check that the online pharmacy is serious and has all the necessary authorizations.
If you have any doubts about the origin of the drug, you should carry out an analysis to certify the presence of the active ingredient and check that its dosage in the drug complies with the indicated content.
Analysis
Atorvastatin is analyzed by liquid chromatography coupled with UV-Visible spectrophotometric detection (HPLC-UV Visible).
Chemical name
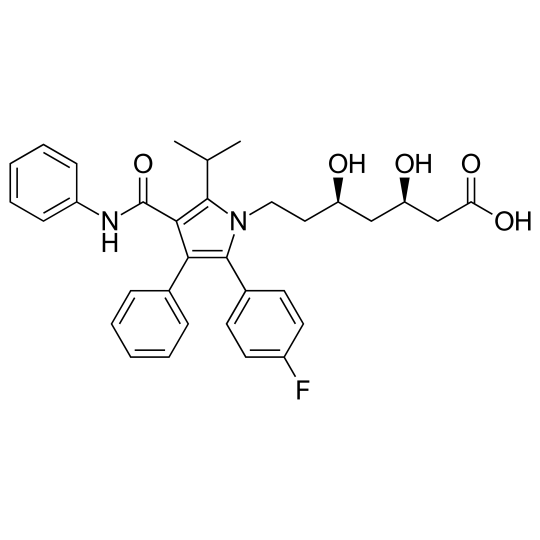
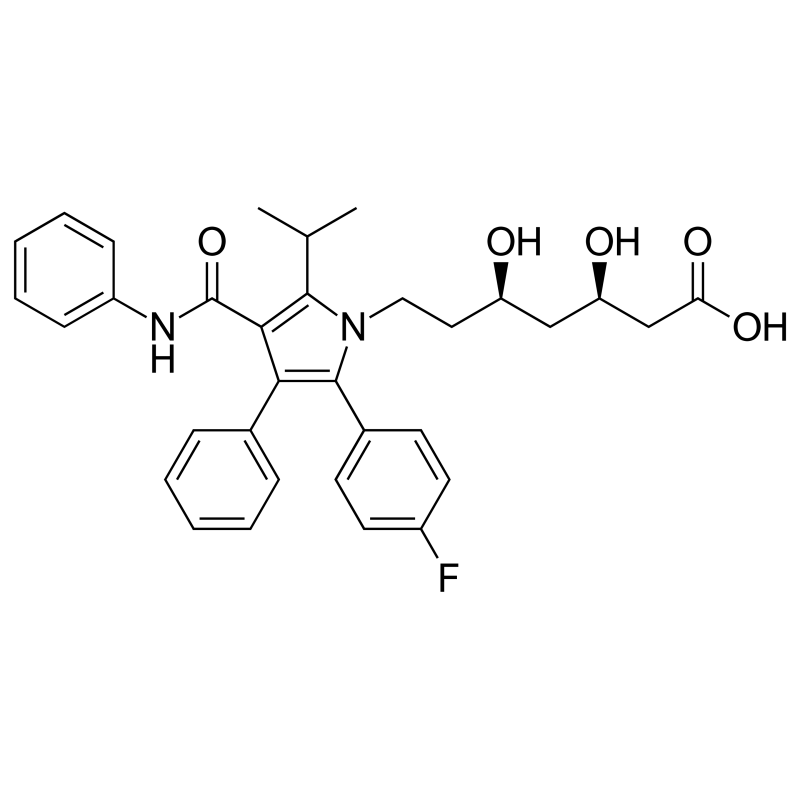
- Chemical name : Calcium (3R,5R)-7-[2-(4-fluorophényl)-3-phényl-4-(phénylcarbamoyl)-5-propylpyrrol-1-yl]-3,5-dihydroxyheptanoate
- Formula : C33H35FN2O5
- CAS number : 134523-00-5
Therapeutic class
Atorvastatin belongs to the statin class (HMG-CoA reductase inhibitors).
Description of therapeutic effects
Atorvastatin is indicated for :
- Reduces LDL ("bad") cholesterol : Blocks a liver enzyme responsible for cholesterol synthesis.
- Increase in HDL ("good") cholesterol: Improves blood lipid balance.
- Prevention of cardiovascular disease: Reduces the risk of myocardial infarction and stroke.
- Treatment of dyslipidemia: Indicated for patients with familial or mixed hypercholesterolemia.
Atorvastatin acts by inhibiting the HMG-CoA reductase enzyme, thus reducing cholesterol production in the liver.
Trade names
- Lipitor
- Tahor
- Totalip
- Numerous generics available
Main manufacturers
- Pfizer
- Sanofi
- Teva Pharmaceuticals
- Mylan
- Sandoz
Specific References
Your drug is analyzed by our expert pharmaceutical analysis laboratory.
The drug to be analyzed must be sent in at least 3 copies in its original packaging (box or vial + blister pack) in order to have all the information.
The drug to be analyzed must be sufficiently protected to avoid any deterioration or damage during transport.
The drug to be analyzed must be accompanied by the completed analysis form.
Documents to download:
The use of counterfeit or illegal medicines (without patent authorisation) presents a proven risk to patients' health:
- Absence of the active ingredient
- Under-dosage of the active ingredient
- Over-dosage of the active ingredient
- Toxic synthetic or degradation impurities
- Presence of another active ingredient
- Toxic excipients
- Presence of toxic residual solvents
- ...
The trade in counterfeit medicines is flourishing and growing exponentially with online trading. The authorities believe that more than half of all medicines sold over the Internet are counterfeit or illegal.