Ampicillin - Penbritin - Unacim Analysis
Laboratory analysis of drugs to certify the presence of Ampicillin and measure the quantity of Ampicillin.
The level of uncertainty of the Ampicillin content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%).
Follow the instructions to send your drug to our laboratory for analysis.
Results within 15 working days of receipt of sample.
Ampicillin is a popular broad-spectrum antibiotic used to treat bacterial infections. Ampicillin is included in the World Health Organization's (WHO) list of essential medicines.
Ampicillone is the active ingredient in Penbritin, produced by Bristol-Myers Squibb (BMS).
Ordering medicines from dubious websites is a health risk. It is essential to check that the online pharmacy is serious and has all the necessary authorizations.
If you have any doubts about the origin of the drug, you should carry out an analysis to certify the presence of the active ingredient and check that its dosage in the drug complies with the indicated content.
Analysis
Ampicillin is analyzed by liquid chromatography coupled with UV-Visible spectrophotometric detection (HPLC-UV Visible).
Chemical name
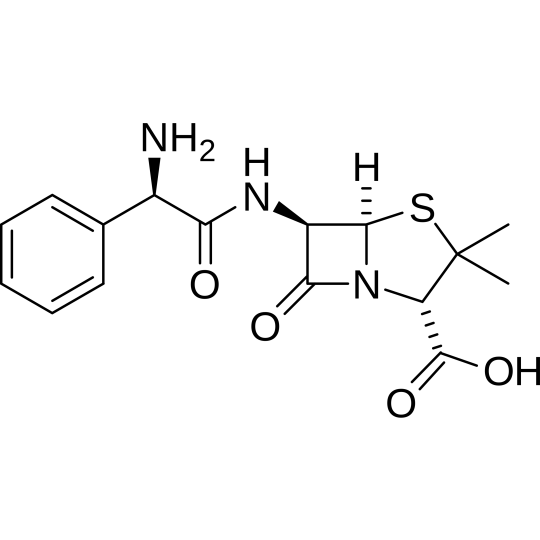
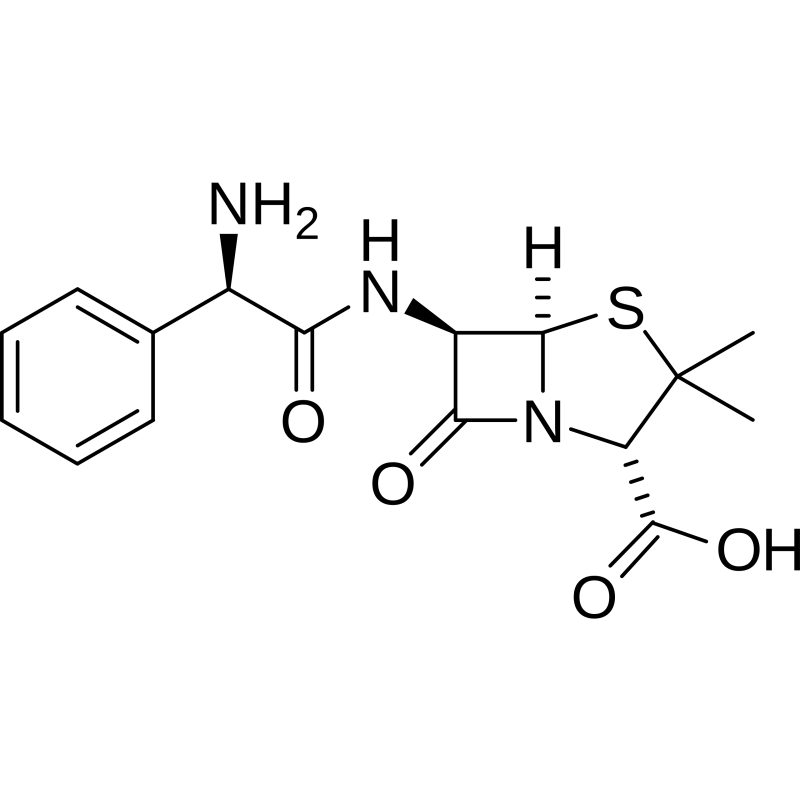
- Chemical name : (2S,5R,6R)-6-[(2R)-2-amino-2-phénylacétamido]-3,3-diméthyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
- Chemical formula : C16H19N3O4S
- CAS number: 69-53-4
Therapeutic class
- Therapeutic class : Antibiotic
- Subclass: Beta-lactam antibiotics, amino penicillins
Description of Therapeutic Effects
Ampicillin is a beta-lactam antibiotic used to treat a wide range of bacterial infections. It is active against many Gram-positive and some Gram-negative bacteria.
- Mechanism of action: Ampicillin acts by inhibiting bacterial cell wall synthesis. It binds to penicillin-binding proteins (PBPs) and blocks the formation of peptidoglycans, which weakens the bacterial cell wall, leading to bacterial lysis and death.
- Use: Ampicillin is indicated for treating respiratory tract infections, urinary tract infections, gastrointestinal infections, ear infections, biliary tract infections, bacterial meningitis and other serious infections caused by susceptible bacteria. It is also used to prevent bacterial endocarditis.
Trade names
Ampicillin is available under various brand names, including :
- Totapen: Used in some countries for oral and injectable formulations.
- Penbritin: Another well-known brand name for ampicillin
- Ampicilline Sandoz: A common generic version
- Omnipen: Marketed for injectable formulations
- Ampicillin Mylan: Another popular generic brand name
Main Manufacturers
The main manufacturers of ampicillin are:
- Bristol-Myers Squibb (BMS): One of the leading producers of ampicillin under the brand name Penbritin.
- Sandoz (division of Novartis): Producer of generic ampicillin.
- Pfizer: Manufacturer of certain ampicillin formulations, notably injectables.
- Mylan: Major supplier of generic versions of ampicillin.
- GlaxoSmithKline (GSK): Produced and marketed ampicillin in the past.
- Sun Pharmaceutical Industries: Manufacturer of generic formulations for global markets.
- Cipla : Producer of ampicillin, particularly for developing countries.
Precautions and side effects
- Precautions: Ampicillin is contraindicated in people allergic to penicillins or other beta-lactam antibiotics. Susceptibility testing is often recommended before initiating treatment to ensure the antibiotic's efficacy. Special monitoring is required in patients with renal or hepatic impairment.
- Common side effects: Nausea, vomiting, diarrhea, rash, oral or vaginal candidiasis.
- Serious side effects: Severe allergic reactions (anaphylaxis), antibiotic-associated colitis, hepatitis, leukopenia (reduced white blood cell count), kidney damage.
Specific References
Your drug is analyzed by our expert pharmaceutical analysis laboratory.
The drug to be analyzed must be sent in at least 3 copies in its original packaging (box or vial + blister pack) in order to have all the information.
The drug to be analyzed must be sufficiently protected to avoid any deterioration or damage during transport.
The drug to be analyzed must be accompanied by the completed analysis form.
Documents to download:
The use of counterfeit or illegal medicines (without patent authorisation) presents a proven risk to patients' health:
- Absence of the active ingredient
- Under-dosage of the active ingredient
- Over-dosage of the active ingredient
- Toxic synthetic or degradation impurities
- Presence of another active ingredient
- Toxic excipients
- Presence of toxic residual solvents
- ...
The trade in counterfeit medicines is flourishing and growing exponentially with online trading. The authorities believe that more than half of all medicines sold over the Internet are counterfeit or illegal.