Quinine - Quinimax Analysis
Laboratory analysis of drugs to certify the presence and measure the quantity of Quinine.
The level of uncertainty of the Quinine content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%).
Follow the instructions to send your drug to our laboratory for analysis.
Results within 15 working days of receipt of sample.
Quinine is a drug used to treat acute malaria attacks caused by Plasmodium falciparum, which is resistant to other antimalarial drugs, and is sometimes used to relieve nocturnal leg cramps.
Quinine is the active ingredient in Quinimax, marketed by the Sanofi laboratory.
Consuming medicines ordered from dubious or illegal websites can be hazardous to your health. It is essential to check that the online pharmacy is serious and has all the necessary authorizations.
If you have any doubts about the origin of the drug, you should carry out an analysis to certify the presence of the active ingredient and check that its dosage in the drug complies with the indicated content.
Analysis
Quinine is analyzed by liquid chromatography coupled with UV-Visible spectrophotometric detection (HPLC-UV Visible).
Chemical name
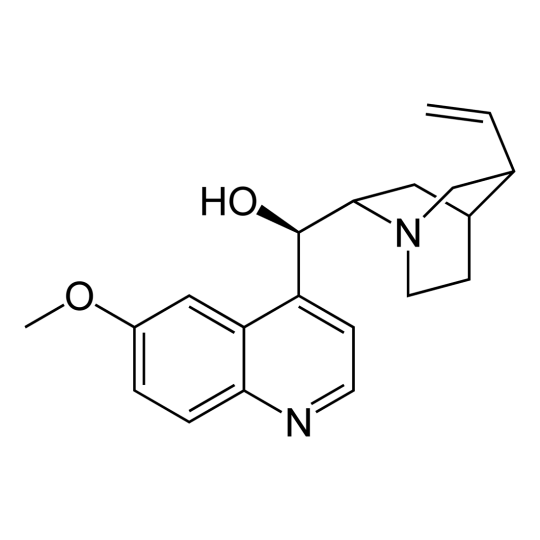
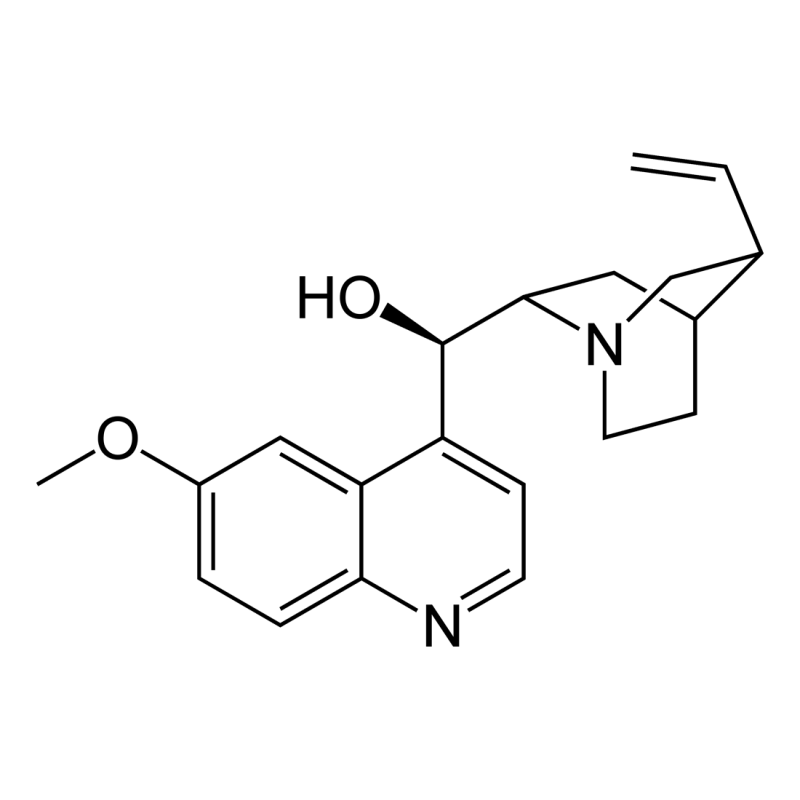
- Chemical name: (R)-[(2S,4S,5R)-5-ethenyl-1-azabicyclo[2.2.2]octan-2-yl] (6-methoxyquinolin-4-yl)methanol
- Chemical formula: C20H24N2O2
- CAS number: 130-95-0
Therapeutic class
- Therapeutic class: antimalarial and antispasmodic.
Description of Therapeutic Effects
Quinine is used for :
- Treatment of acute attacks of severe malaria :
- Active against resistant strains of Plasmodium falciparum.
- Treatment of nocturnal leg cramps (off-label use in certain cases).
Mechanism of action :
- Quinine interferes with heme detoxification in the parasite, leading to the accumulation of toxic products and death.
Trade names
- Quinimax (Sanofi)
- Quinine sulfate (generics).
Main Manufacturers
- Sanofi: Leading manufacturer of Quinimax.
- Cipla: Producer of generics in developing countries.
- Bayer: Supplier in certain historical markets.
- Novartis: Manufacturer of specific formulations.
Specific References
Your drug is analyzed by our expert pharmaceutical analysis laboratory.
The drug to be analyzed must be sent in at least 3 copies in its original packaging (box or vial + blister pack) in order to have all the information.
The drug to be analyzed must be sufficiently protected to avoid any deterioration or damage during transport.
The drug to be analyzed must be accompanied by the completed analysis form.
Documents to download:
The use of counterfeit or illegal medicines (without patent authorisation) presents a proven risk to patients' health:
- Absence of the active ingredient
- Under-dosage of the active ingredient
- Over-dosage of the active ingredient
- Toxic synthetic or degradation impurities
- Presence of another active ingredient
- Toxic excipients
- Presence of toxic residual solvents
- ...
The trade in counterfeit medicines is flourishing and growing exponentially with online trading. The authorities believe that more than half of all medicines sold over the Internet are counterfeit or illegal.