Artesunate - Artesun - Artem - Arinat - Malacef Analysis
Laboratory analysis of drugs to certify the presence of Artesunate and measure the quantity of Artesunate.
The level of uncertainty of the Artesunate content analysis depends on the declination selected (Standard <20%, Precise <10% or Regulatory <5%).
Follow the instructions to send your drug to our laboratory for analysis.
Results within 15 working days of receipt of sample.
Artesunate is an antiparasitic drug used to treat malaria. Artesunate is included in the World Health Organization (WHO) list of essential medicines.
Artesunate is the active ingredient in Arinat marketed by DAFRA PHARMA and ASAQ marketed by Sanofi.
Ordering medicines from dubious websites is a health risk. It is essential to check that the online pharmacy is serious and has all the necessary authorizations.
If you have any doubts about the origin of the drug, you should carry out an analysis to certify the presence of the active ingredient and check that its dosage in the drug complies with the indicated content.
Analysis
Artesunate is analyzed by liquid chromatography coupled with UV-Visible spectrophotometric detection (HPLC-UV Visible).
Chemical name
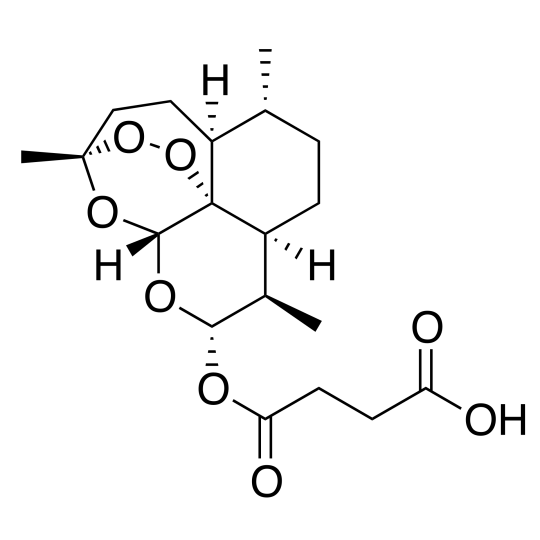
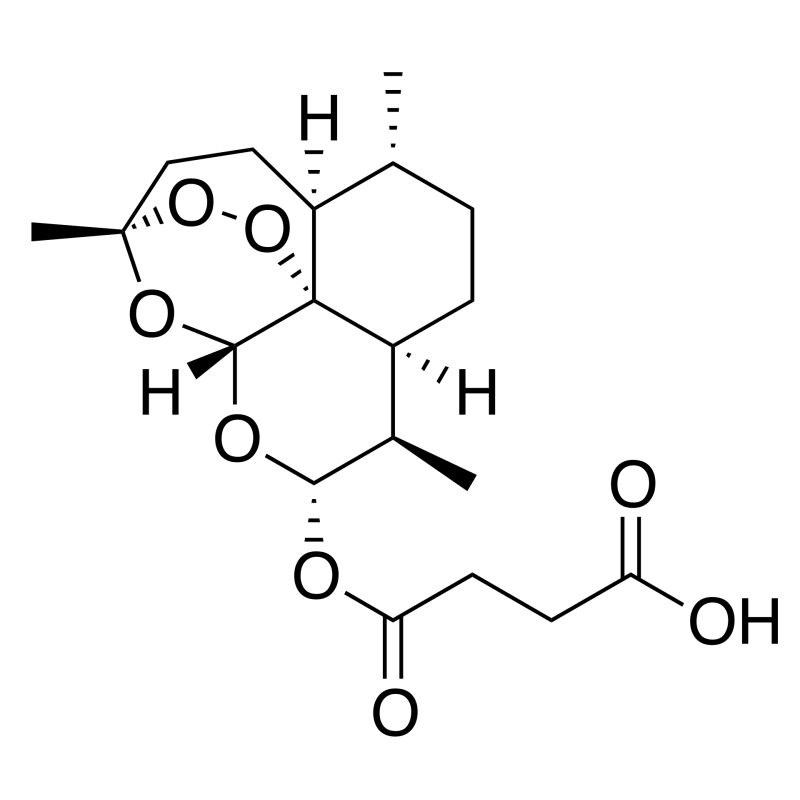
- Chemical name : (3R,5aS,6R,8aS,9R,10S,12R,12aR)-Decahydro-10-oxo-3,6,9-triméthyl-3,12-époxy-12H-pyrano[4,3-j]-1,2-benzodioxépine-4-yl methylsuccinate
- Formule chimique : C19H28O8
- Numéro CAS : 88495-63-0
Therapeutic class
- Therapeutic class: Antimalarial
- Subclass: Artemisinin derivatives
Description of Therapeutic Effects
Artesunate is a highly effective anti-malarial agent derived from artemisinin, used primarily in the treatment of severe malaria, notably caused by Plasmodium falciparum. It is particularly crucial in situations where malaria is life-threatening.
- Mechanism of action: Artesunate acts rapidly to kill malaria parasites by producing free radicals in the presence of iron in the parasite. This causes irreversible damage to the parasite's membranes and other vital components. Artesunate is transformed in the body into its active metabolite, dihydroartemisinin (DHA), which has a powerful anti-parasitic action.
- Use: It is used as an emergency treatment for severe malaria, either intravenously or intramuscularly. It can also be administered orally for the treatment of uncomplicated malaria, but is generally combined with another antimalarial drug to prevent relapses and reduce the risk of resistance.
Trade names
Artesunate is sold under various trade names, including :
- Artesun: Trade name commonly used for injectable formulations of artesunate.
- ASAQ: A combination of artesunate and amodiaquine, used in some countries for the treatment of uncomplicated malaria
- Coarsucam: Another combination of artesunate and amodiaquine
- Artesunate Winthrop: A product manufactured by Sanofi for the international market
- Artem: Another trade name for artesunate, particularly in developing countries
Main Manufacturers
The main manufacturers of artesunate are :
- Guilin Pharmaceutical: One of the world's leading manufacturers of artesunate, certified by the WHO for the production of malaria treatments.
- Sanofi: Producer of Artesunate Winthrop and the ASAQ combination, deeply involved in the fight against malaria.
- Cipla: Supplier of artesunate formulations for several markets in Africa and Asia.
- Ipca Laboratories: Indian producer of artesunate, recognized for its worldwide exports.
- Ajanta Pharma: Manufacturer of artesunate tablets and injections for health programs in endemic regions.
- Mylan: Producer of anti-malarial drugs, including products containing artesunate.
Precautions and side effects
- Precautions: Artesunate is generally well tolerated, but should be administered with caution in patients with a history of hypersensitivity to artemisinin derivatives. Regular monitoring is recommended for possible hematological side effects, including delayed anemia.
- Common side effects: Abdominal pain, nausea, dizziness, headache, weakness, injection site reactions.
- Serious side effects: Delayed hemolytic anemia, severe allergic reactions (anaphylactic shock), liver disorders.
Specific References
Your drug is analyzed by our expert pharmaceutical analysis laboratory.
The drug to be analyzed must be sent in at least 3 copies in its original packaging (box or vial + blister pack) in order to have all the information.
The drug to be analyzed must be sufficiently protected to avoid any deterioration or damage during transport.
The drug to be analyzed must be accompanied by the completed analysis form.
Documents to download:
The use of counterfeit or illegal medicines (without patent authorisation) presents a proven risk to patients' health:
- Absence of the active ingredient
- Under-dosage of the active ingredient
- Over-dosage of the active ingredient
- Toxic synthetic or degradation impurities
- Presence of another active ingredient
- Toxic excipients
- Presence of toxic residual solvents
- ...
The trade in counterfeit medicines is flourishing and growing exponentially with online trading. The authorities believe that more than half of all medicines sold over the Internet are counterfeit or illegal.